 
Although J&J is at least two months behind Pfizer (NYSE: PFE) and Moderna (NASDAQ: MRNA) who have already started to roll out their vaccines, J&J’s shot could be much sought after if it proves as safe and effective as rivals, considering that it likely requires only a single dose, unlike rival vaccines which require two shots to be given a few weeks apart. If all goes well, J&J could apply for emergency use approval from the U.S. are likely to allow it to gather the data it needs with fewer volunteers. The company has completed enrollment on the trial, with 45,000 people for the trial, below its initial target of 60,000, although this is unlikely to make a difference as higher rates of Coronavirus infections in the U.S. 
Johnson & Johnson (NYSE:JNJ) is expected to report interim data from the phase 3 trial of its Covid-19 vaccine candidate this month, providing insight into its safety and efficacy. What’s Happening With Johnson & Johnson’s Covid-19 Vaccine? See our indicative theme of Covid-19 Vaccine stocks – which includes a diverse set of U.S.-based pharma and biotech companies developing Covid vaccines. As of last September, the company said that it had plans to deliver over a billion doses by the end of 2021. While J&J was expected to deliver 12 million doses by the end of February and 100 million by the end of June, it has reportedly fallen behind these goals by as much as two months. Separately, there have been reports that the company is seeing some manufacturing delays for the vaccine. More conclusive efficacy data from J&J’s phase 3 study which has 45,000 volunteers is expected in the coming weeks with the company likely to proceed with emergency approval if the results are favorable. For perspective, the Pfizer and Moderna vaccines, which are being rolled out in the U.S. Based on these early-stage findings the company expects the vaccine to be more than 70% effective, noting that efficacy could reach “very high levels”. Based on data from the phase 1/2 trials of 805 participants, published on Wednesday, the vaccine generated a long-lasting immune response with 90% of participants generating neutralizing antibodies against the Coronavirus. Here’s a quick rundown on the recent developments for the vaccine. The Pfizer vaccine must be stored at minus 94 degrees Fahrenheit.Johnson & Johnson (NYSE:JNJ) is developing one of the most closely watched Covid-19 vaccines, considering that it is a single-dose shot that should be relatively easy to distribute. The Johnson & Johnson vaccine is seen as critical to fighting the pandemic in part because it requires only one dose - and doesn’t need to be stored at sub-zero temperatures.īy contrast, the Pfizer and Moderna vaccines require two separate doses. The company, which has pledged to release 100 million doses by late June, will seek emergency FDA approval for its COVID-19 vaccine candidate later this month. On Monday, Operation Warp Speed co-director Moncef Slaoui hinted there may be a delay in Johnson & Johnson’s initial goal of 12 million, saying “single-digit million” doses would likely be delivered by the second half of February. “Operation Warp Speed is working with Johnson & Johnson to scale up and maximize manufacturing of the Janssen vaccine … Making projections at this time is premature.” “We remain in active discussions with regulators, including on the approval and validation of our manufacturing processes,” the rep said. It may now take Johnson & Johnson until late April to catch up with that original projection, a person briefed on the matter told the outlet.īut a rep for the pharmaceutical giant denied it was experiencing a production slowdown - saying the company is “confident in our ability to meet our 2021 supply commitments.” The pharmaceutical giant, which vowed to make 12 million of the single-dose jabs available by the end of February, warned health officials that the firm has fallen behind, according to Politico. 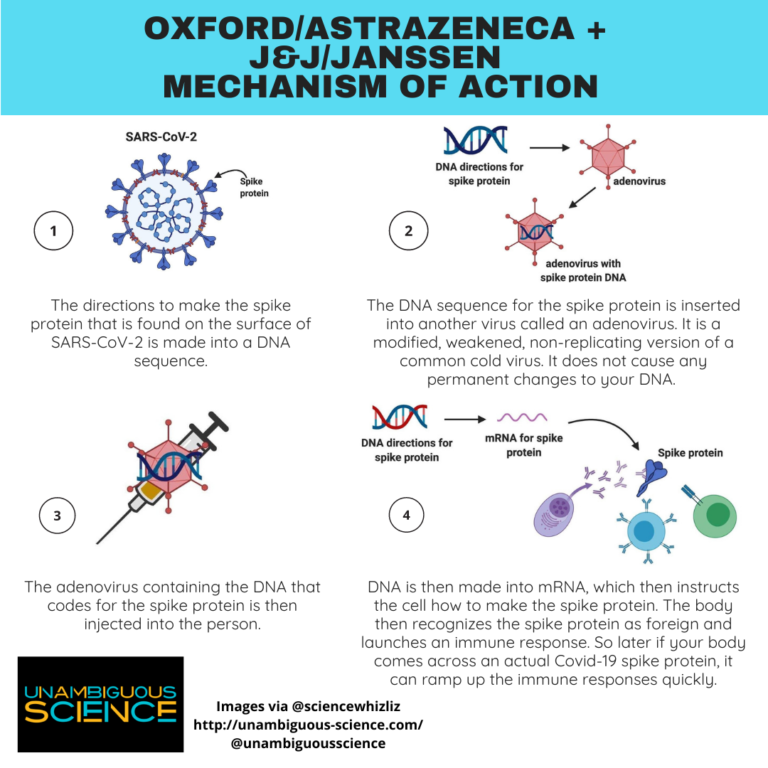
Johnson & Johnson’s COVID-19 vaccine has hit a production snag - potentially delaying the delivery of millions of life-saving shots by two months, according to a report Wednesday. NY’s Hochul quietly scraps $64M Excelsior COVID vax app due to $200K monthly costs and fact no one uses it anymore Biden admin thinks it has divine right to nix conservatives’ free speech rightsĬDC boss’ utterly laughable exit warning on politicized ‘science’Īgeism in NYC is a serious issue with 29% of NYers believing seniors shouldn’t work: poll 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
